
Sciencare is a patient-focused innovator in addiction treatment, dedicated to bringing our long-acting implant technologies to patients worldwide. Through strategic partnerships, we aim to address the global challenge of substance abuse.
Since its establishment,Since its establishment, Sciencare has been committed to addressing the global challenge of psychoactive substance addiction.We have built an innovative drug delivery R&D platform centered on our proprietary long-acting implant technology.
Through years of R&D innovation accumulation, Sciencare has formed a drug delivery technology platform on long-acting implants technology, and focuses on innovative applications in addiction treatment,and central nervous system disease treatment, found multiple implant pipelines.
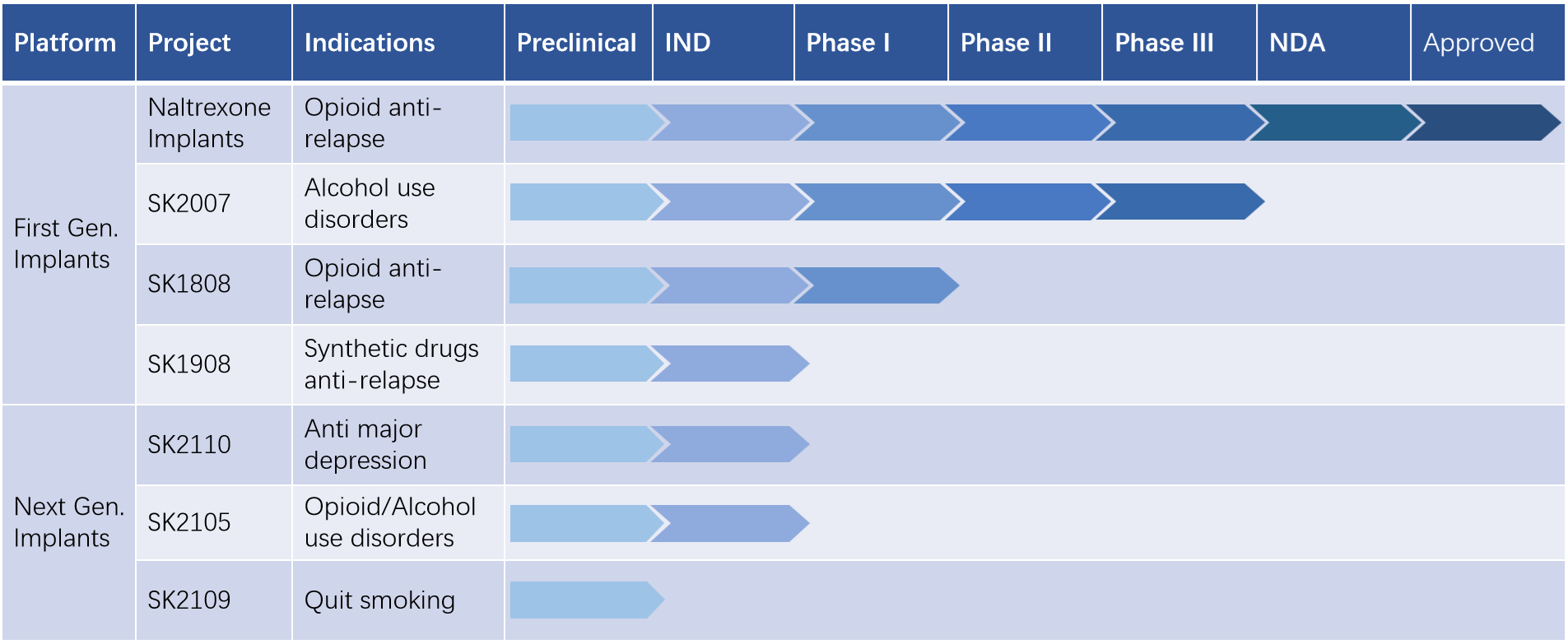
SK2007 is the first ultra long acting abstinence medication that lasts up to 150 days in the world. A single dose of the medication can effectively reduce alcohol consumption. It has completed Phase II clinical trials and Phase III clinical trials is in progress.
SK1808 is a registered opioid anti relapse indication product of naltrexone hydrochloride implant in the United States, which complies with FDA regulatory requirements. In 2025, it was granted implied permission by the FDA to conduct Phase I clinical trials in the United States.
SK1908 implant is used for anti relapse treatment of new drugs such as methamphetamine, and is also the world's first drug used for preventing relapse of marijuana addiction. It can be released for 3 months after one implantation and is about to be declared for IND.
SK2110 implant, the first long-acting formulation for treating severe depression,The IND application is about to be submitted.
SK2105 implant, prevention of relapse in patients with strong addiction to fentanyl drugs, and treatment of alcohol use disorders,The IND application is about to be submitted.
SK2109 implant, the world's first long-acting smoking cessation implant, with a continuous release of the drug for 3 months after implantation.